| Hint | Food | 맛과향 | Diet | Health | 불량지식 | 자연과학 | My Book | 유튜브 | Frims | 원 료 | 제 품 | Update | Site |
|
조절물질 ≫ 신경전달, 호르몬 식물 : 트리고넬린 trigonellin 식물호르몬 - Abscisic Acid, Auxin, Brassinosteroids - Cytokinins, Ethylene, Gibberellins - Jasmonates, Salicylic Acid, Strigolactones - 트리고넬린 트리고넬린은 여러 식물과 동물에도 존재하는데 커피에는 2% 정도 들어 있다. 트리고넬린은 니코틴산으로부터 만들어지는데 이 반응이 가역적이라 식물에 따라 트리고넬린이 니코틴산의 저장 수단으로 이용되기도 한다. 그리고 트리고넬린은 생각보다 중요한 식물 대사 조절물질이다. 세포 주기를 조절하고, 산화적 스트레스에 대한 신호 전달을 하며, 삼투압 조절도 한다. 그리고 잎의 폐쇄 여부 조절, 수면주기 조절, 발아 조절, 자외선 스트레스로부터 보호, 염 스트레스로부터 보호 그리고 커피씨앗 발아 동안에 보존의 역할도 한다. 로스팅 시 일부는 향기 물질이 된다. TRIGONELLINE: A DIVERSE REGULATOR IN PLANTS Trigonelline (N-methyl nicotinamide) first gained the attention of plant biologists because it proved to be very effective in inducing G2 arrest in the root apices of many plant species. Although trigonelline, a metabolite of nicotinamide, was put forth as a possible plant hormone over 20 years ago (Evans and Tramontano, 1981), this idea was to gain little acceptance in the ensuing years. Meanwhile, however, evidence from diverse quarters has slowly been accumulating that indicates that the effects of trigonelline are not just limited to cell cycle regulation: Trigonelline appears to be a regulator of sundry other functions in plants. This month's The Hot and the Classic summarizes the state of knowledge concerning the regulatory functions of trigonelline in plants. Cell Cycle Regulation Evans et al. (1979) identified trigonelline as the substance in pea (Pisum sativum) cotyledons that promoted G2 arrest in root and shoot meristems. Concentrations of trigonelline as low as 10−7 m were found to be effective. Trigonelline is present in ungerminated seeds and is transported to other parts of the seedling during early germination. The fact that added trigonelline can replace cotyledons in promoting G2 arrest, and that the trigonelline levels in planta were highly correlated with the proportion of cells arrested in G2, led to the suggestion that trigonelline may be a natural plant hormone (Evans and Tramontano, 1981). More recent evidence suggests that trigonelline may act as a cell cycle regulator by preventing the ligation of replicons during the S-phase of the cell cycle (Mazzuca et al., 2000). Mean replicon size was determined to be about 2.5-fold longer in lettuce (Lactuca sativa) seedlings treated with 3 mm trigonelline than in controls. Trigonelline also resulted in a lengthening of both the S-phase and the cell cycle and to a decrease in primary root elongation. Hence, Mazzuca et al. (2000) proposed that replicon inactivation may underlie the protracted S-phase and inhibition of growth. Trigonelline treatment also resulted in a 1.6-fold increase in fork rate compared with the control. The faster fork rate in the larger replicons is in accord with the highly significant positive relationship that has been established between fork rate and replicon size for various unrelated higher plants. Mazzuca et al. (1997) also noted that trigonelline treatments caused the nucleoli of plant cell nuclei to become very large and to undergo heavy labeling with radioactive thymidine. These changes were clearly related to the presence of trigonelline as the size of the nucleoli rapidly diminished following a recovery period in water. Since the modifications of the nucleoli detected in treated roots were accompanied by changes in the protein pattern, the results indicate that trigonelline may exert its role through synthesis of new specific proteins. Nodulation In addition to the flavonoids exuded by many legumes as signals to their rhizobial symbionts, alfalfa (Medicago sativa) releases trigonelline into the rhizosphere. Trigonelline specifically activates the expression in Rhizobium meliloti of a class of genes (trc genes) that are apparently involved in trigonelline catabolism (Boivin et al., 1990). The authors suggest that trigonelline may be used as a nutrient source by the bacteria during all stages of the symbiotic relation, including in the rhizosphere, and in the nodules of the host plant. Althoughtrc mutants produce normal appearing nodules under laboratory conditions, the authors speculate that under field conditions, the presence of these catabolic genes may confer upon certain strains a selective advantage for the colonization of the rhizosphere or in the development of the plant infection or both. Oxidative and UV Stresses Berglund (1994) has put forth the interesting hypothesis that nicotinamide is an important part of the signal transduction chain involved in the response of plant cells to conditions that cause DNA strand breakage, especially in connection with oxidative stress. In stressed cells, nicotinamide is released as a result of the activity of the nuclear enzyme poly(ADP-Rib) polymerase (PADPRP). PADPRP is activated by various types of stress that cause DNA strand breakage, including oxidative stress, UV stress, and mutagens. The activation of PADPRP does not occur at the level of the gene, but through the interaction of PADPRP with damaged DNA. PADPRP synthesizes polymers of ADP-Rib that become attached to various DNA-associated proteins. The ADP-Rib consumed in this process comes from NAD, which is thus degraded with the release of nicotinamide. Thus, Berglund proposes that during those types of stress that cause an increased frequency of strand breaks in DNA, there is a rapid increase in nicotinamide (and consequently trigonelline) levels within plant cells. Berglund proposes that nicotinamide and trigonelline may serve as potent inducers of defensive metabolism in plants, including glutathione metabolism, and the accumulation of secondary defense compounds. Indeed, Kalbin et al. (1997) found that strong UV-B irradiation caused marked increases in the levels of nicotinamide, trigonelline, and total oxidized glutathione in pea leaves. They concluded that elevated levels of nicotinamide and trigonelline do occur in response to UV-B, but only at UV-B doses high enough to cause oxidative stress (see also Berglund et al., 1996). Thus, nicotinamide and/or its metabolites (including trigonelline) may function as signal transmitters in the response of plants to oxidative stress, and poly(ADP-Rib) polymerase may play an important role in the induction of defensive metabolism. DNA Methylation It has been suggested that the physiological effects of trigonelline and other quaternary ammonium compounds in plants could occur at the level of DNA methylation (Kraska and Schönbeck, 1993). Trigonelline, choline, and betaine show a hypo-methylating effect in plants. In the case of trigonelline,Berglund (1994) proposes that the deamidation of nicotinamide to nicotinic acid followed by the methylation of nicotinic acid to trigonelline, may consume S-adenosyl-Met, which is the methyl donor employed when DNA is methylated. Because DNA methylation is generally linked to DNA replication, DNA demethylation may play a role in mediating the effects of trigonelline on the cell cycle. Salt Stress In response to excess salt, many plants accumulate osmoregulators such as Gly betaine, Pro, and trigonelline to prevent water loss. Tramontano and Jouve (1997) found that alfalfa plants undergo a 5-fold increase in Pro and a 2-fold increase in trigonelline after salt-stress. Further experiments examined whether other known osmoregulators (e.g. Pro and Gly betaine) could affect the cell cycle parameters in cultured root meristems of peas in a manner similar to trigonelline. At concentrations of 10−4 to 10−7 m, trigonelline induced an accumulation of G2 nuclei, whereas Pro was ineffective and Gly betaine only slightly effective in promoting G2 nuclei accumulation. These results confirm that the cell cycle effects of trigonelline are indeed specific and that trigonelline may play a role as an osmoregulator in salt-stressed plants, an idea strengthened by the observation by Shomerilan, Jones, and Paleg (1991) that trigonelline, like Pro, increases the in vitro thermal and salt stability of pyruvate kinase. Nyctinasty The laboratory of M. Ueda has been extremely prolific in identifying factors from leaf extracts that are effective in inducing leaf closure in various species that undergo pronounced sleep movements. Trigonelline was isolated from Aeschynomene indica as a bioactive substance for nyctinasty (Ueda, Niwa, and Yamamura, 1995). The compound was quite effective for leaf closing of this species at 0.1 μm in the daytime, but not for the nyctinastic species Cassia mimosoides and Mimosa pudica. It competed with indole-3-acetic acid, which is effective in leaf opening. These results suggest that trigonelline may be involved in the circadian rhythm of A. indica. 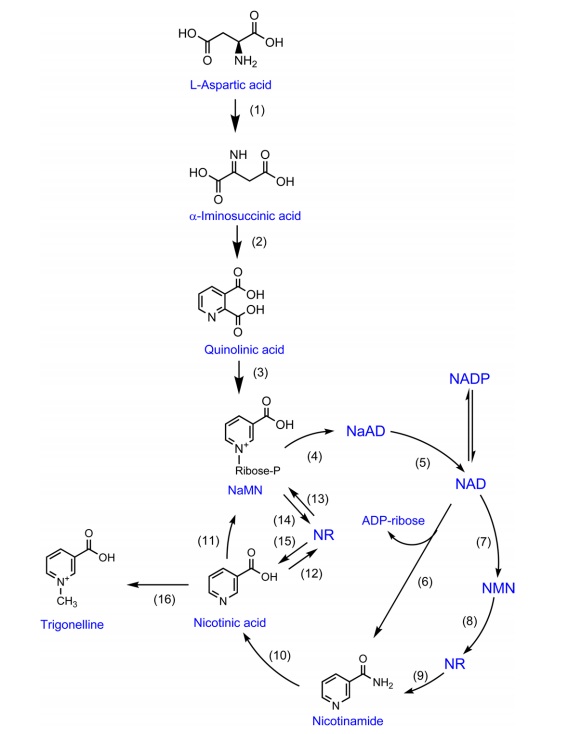 |
||||
|
|
|||